Insights / Blog / EDC
eCRFs in Clinical Trials: Understanding Structure, Challenges, and the Role of AI
- Abriti Rai
- February 28, 2026

On this Page
- Summary
- What Is a Case Report Form in Clinical Trials?
- Evolution of eCRF Clinical Trials
- Key Components of an eCRF
- How eCRFs Work in Clinical Trials
- Who Uses eCRF Systems in Clinical Trials?
- Benefits of eCRF in Clinical Trials
- Considerations While Designing an eCRF
- eCRF vs EDC: How eCRFs Fit Into Modern EDC Systems
- Challenges in eCRF Clinical Trials
- How AI Is Enhancing eCRF Design and Standardization
- Conclusion
- Summary
- What Is a Case Report Form in Clinical Trials?
- Evolution of eCRF Clinical Trials
- Key Components of an eCRF
- How eCRFs Work in Clinical Trials
- Who Uses eCRF Systems in Clinical Trials?
- Benefits of eCRF in Clinical Trials
- Considerations While Designing an eCRF
- eCRF vs EDC: How eCRFs Fit Into Modern EDC Systems
- Challenges in eCRF Clinical Trials
- How AI Is Enhancing eCRF Design and Standardization
- Conclusion
Summary
eCRF in clinical trials is the electronic case report form used to record participant data in a structured, digital format. What began as a simple replacement for paper CRFs has evolved into an intelligent data foundation, supporting automation, integration, and data quality across the clinical research lifecycle.
A well-designed eCRF ensures accurate data, smoother monitoring, compliance with regulations, and better trial results. Understanding what a case report form is in clinical trials and how its electronic evolution reshapes trial operations is essential for sponsors, CROs, data managers, and clinical operations teams alike.
What Is a Case Report Form in Clinical Trials?
A case report form (CRF) is a structured document used to systematically collect protocol-specified data for each trial participant. It captures clinical, laboratory, safety, and efficacy information required for statistical analysis, regulatory submission, and overall trial evaluation.
Core Purpose of a CRF
- Standardize data collection across sites to reduce variability and ensure comparable datasets.
- Ensure protocol adherence by guiding investigators to capture required data points accurately.
- Enable accurate statistical analysis for robust study outcomes.
- Support regulatory inspections and audits by maintaining clear, traceable records of participant data.
Transition from Paper CRF to eCRF
While paper CRFs laid the foundation for standardized data collection, their manual nature introduced delays, errors, and resource-intensive monitoring - challenges that the transition to electronic case report forms (eCRFs) was designed to overcome.
Aspect | Paper CRF | eCRF |
Data Entry | Manual | Electronic |
Error Detection | Retrospective | Real-time |
Query Resolution | Delayed | Automated |
Monitoring | On-site heavy | Risk-based / remote |
Scalability | Limited | High |
Evolution of eCRF Clinical Trials
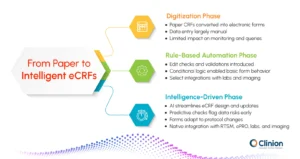
The journey of eCRF clinical trials can be broadly categorized into three phases:
Digitization Phase:
The initial phase involved converting paper CRFs into electronic formats with minimal automation.
At this stage, eCRFs largely mirrored paper forms, with manual data entry, retrospective error detection, and limited operational efficiency. While accessibility improved, digital potential remained underutilized.
Rule-Based Automation Phase:
This phase introduced edit checks, validations, and conditional logic.
eCRFs enabled real-time error detection, automated queries, and more efficient monitoring. Operational efficiency improved, transcription errors decreased, and sponsors gained faster study insights, although adaptability to protocol changes remained limited.
Intelligence-Driven Phase:
The latest phase integrates AI in eCRF design, enabling adaptive workflows, predictive data quality controls, and intelligent form recommendations.
Modern eCRFs anticipate potential errors, adapt dynamically to protocol amendments, and integrate seamlessly with RTSM, ePRO, labs, and imaging systems, enhancing data quality and site usability.
Modern clinical trials increasingly demand eCRFs that are:
- Protocol-aware: Automatically align with trial protocols and amendments.
- Adaptive to amendments: Dynamically update forms as study requirements change.
- Integrated: Smoothly connect with RTSM, ePRO, labs, and imaging systems.
- User-friendly: Optimized for real-world usability at clinical sites, reducing training burden and data entry errors.
This evolution illustrates how eCRF automation has moved from simple digitization to intelligence-driven systems, laying the foundation for faster, more accurate, and more compliant clinical trials.
The Rise of Global Libraries and Built-in Compliance (h3)
With the shift towards eCRFs, the industry is also embracing pre-built global libraries that streamline data collection, ensure regulatory compliance from the outset, and contain standardized data points adhering to CDASH guidelines. This eliminates the need to recreate forms from scratch for each study, saving time, ensuring consistency across trials, and expediting study initiation with compliance built right in.
Key Components of an eCRF
An electronic case report form (eCRF) is only as effective as its components. Each element ensures accurate, standardized, and auditable data collection while enabling smooth downstream analysis. Understanding these building blocks is crucial for creating forms that support modern clinical trials.

Below are the essential components of an eCRF:
- Form Label
The form label provides a concise title describing the type of data captured, helping site staff quickly identify the form within the study’s visit schedule. - Group Label
Groups organize related questions within a form, allowing recurring sets (e.g., vital signs at multiple visits) and improving clarity for clinical staff. - Item Label
This is a short, precise prompt directing the user to enter a specific piece of information, such as “Serum Creatinine Level” or “Participant Ethnicity.” - Item Description or Hint
Supplementary text that guides the site user on how to enter the data correctly, without cluttering the item label itself. - Field
The space or input area where the user enters data. Fields can be simple text boxes, checkboxes, radio buttons, or dropdown menus. - Value
The actual input provided by the user, or a system-generated value calculated from other fields (e.g., BMI automatically derived from height and weight). - Choice Label
Represents selectable options within a field (e.g., “Yes,” “No”), which correspond to a standardized value stored in the database. - Data Validation / Edit Checks
Automated rules that flag invalid, missing, or inconsistent entries, enabling real-time query resolution and enhancing data integrity. - Pre-fabricated, Standardized Forms
Many eCRFs leverage pre-built templates aligned with standards like CDASH, MedDRA, and WHO-DD, ensuring uniformity and regulatory compliance across multiple studies.
By carefully assembling these components, an eCRF becomes a reliable tool that captures high-quality data while minimizing errors and administrative burden on sites.
How eCRFs Work in Clinical Trials
Electronic case report forms (eCRFs) enable precise, compliant, and efficient data capture in clinical trials. Each step in the process is critical to maintaining data integrity, ensuring readiness for analysis, and supporting regulatory submission.
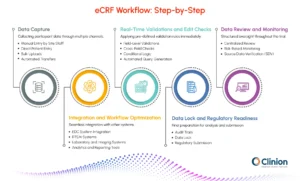
1. Data Capture
eCRFs collect participant data through multiple channels:
Manual Entry by Site Staff:
Clinical research coordinators or investigators enter participant information directly into the eCRF during visits.
Direct Patient Entry:
Participants can provide data through ePROs or patient portals integrated with the eCRF.
Bulk Uploads:
Lab results, imaging data, or other datasets can be uploaded in batches for efficiency.
Automated Transfers:
Integration with external systems (EHRs, laboratory information systems, imaging databases) allows direct data flow into the eCRF, reducing errors and delays.
2. Real-Time Validations and Edit Checks
Once data is entered, the eCRF applies predefined validation rules:
Field-Level Validations:
Ensure values are in acceptable ranges or formats (e.g., numeric ranges, date formats).
Cross-Field Checks:
Confirm consistency between related fields (e.g., adverse event onset date must be before resolution date).
Conditional Logic:
Display relevant fields only when certain criteria are met, reducing unnecessary data entry.
Automated Query Generation:
Any violations trigger queries for immediate resolution by site staff, minimizing downstream corrections.
3. Data Review and Monitoring
eCRFs enable structured oversight throughout the trial:
Centralized Review:
Clinical data managers monitor incoming data for completeness, accuracy, and adherence to protocol.
Risk-Based Monitoring:
eCRFs highlight sites or participants with unusual patterns, enabling targeted interventions rather than broad, resource-heavy checks.
Source Data Verification (SDV):
Monitors can remotely compare eCRF entries against source documents, supported by secure access and audit trails.
4. Integration and Workflow Optimization
Modern eCRFs integrate seamlessly with other trial systems:
EDC System Integration:
eCRFs function as the primary interface for structured data entry within an EDC system, enabling centralized storage, standardized workflows, and secure access for all stakeholders.
RTSM Systems:
Ensures randomization and supply assignments are aligned with collected data.
Laboratory and Imaging Systems:
Automatically imports test results to reduce manual transcription errors.
Analytics and Reporting Tools:
Data is structured for immediate use in interim analyses, safety reporting, and regulatory submissions.
5. Data Lock and Regulatory Readiness
Audit Trails:
Every change is logged with timestamps, user details, and reason for edit, ensuring transparency.
Data Lock:
Once validated and reviewed, datasets are locked for statistical analysis.
Regulatory Submission:
Standardized and structured data support seamless submission to authorities like the FDA or EMA
This stepwise approach ensures that every data point captured via eCRFs is verified, queries are resolved promptly, and the dataset is immediately ready for monitoring, analysis, and regulatory submission, minimizing delays and maximizing trial integrity.
Who Uses eCRF Systems in Clinical Trials?
Electronic case report forms (eCRFs) are used by multiple stakeholders, each with distinct roles that influence system design and usability.
Site Coordinators & Investigators
Enter participant data, record observations, lab results, and adverse events. Clear instructions and validations reduce errors and speed data entry.
Clinical Data Managers (CDMs)
Oversee data quality, resolve discrepancies, manage queries, and ensure protocol compliance. Dashboards and automated checks help detect issues in real time.
Monitors (CRAs)
Perform source data verification and compliance checks. Modern eCRFs support risk-based and remote monitoring, reducing reliance on on-site visits.
Sponsors & Medical Reviewers
Analyze trends, safety signals, and trial progress. Structured datasets and integrated analytics enable confident regulatory reporting.
Benefits of eCRF in Clinical Trials
Effectively implemented eCRFs provide tangible operational efficiencies and scientific advantages, accelerating trial timelines while enhancing data quality and compliance.
Key Benefits
Accelerated study startup through reusable form libraries:
Pre-built eCRFs and standardized templates reduce CRF development time and enable faster trial initiation.
Minimized manual errors and rework:
Automated validations and conditional logic prevent data entry mistakes, improving overall data quality.
Real-time visibility into trial data:
Instant access to participant information allows sponsors, monitors, and data managers to track progress and identify trends immediately.
Strengthened adherence to regulatory standards:
Structured, standardized, and CDISC-compliant eCRFs facilitate smoother audits, inspections, and submissions.
Lower monitoring and operational costs:
Integrated workflows and remote monitoring capabilities reduce the need for frequent site visits, optimizing resource allocation.
Operational Impact of eCRF
Trial Phase | Benefit |
Study Setup | Faster CRF development and deployment using standardized templates and libraries |
Data Entry | Reduced errors and increased consistency with automated edit checks and field validations |
Monitoring | Fewer on-site visits required through remote monitoring and risk-based oversight |
Data Lock | Streamlined timelines for analysis and reporting with validated, ready-to-submit datasets |
These benefits depend heavily on thoughtful eCRF design, intelligent automation, and seamless integration with other trial systems such as RTSM, ePRO, lab, and imaging platforms. Properly executed, eCRFs accelerate trial timelines, enhance data integrity, and ensure actionable insights are available when they are needed most.
Considerations While Designing an eCRF
Designing an eCRF goes beyond technical setup; it must fit real-world trial workflows and evolving protocols.
Key Considerations
- Protocol complexity & visit schedule: Align forms with study visits to capture all required data efficiently.
- Site usability & workload: Keep navigation intuitive, instructions clear, and minimize free-text fields.
- Protocol amendments: Ensure forms can adapt quickly to study changes.
- System integration: Connect seamlessly with EDC, RTSM, ePRO, labs, and imaging.
- Regulatory compliance: Follow CDISC and other standards for audits and submissions.
Pitfalls to Avoid
- Over-validation increases site burden
- Redundant or duplicate data collection
- Misalignment with statistical analysis needs
- Rigid, inflexible designs
Thoughtful eCRF design ensures efficiency, accuracy, and supports AI in eCRF and eCRF automation.
eCRF vs EDC: How eCRFs Fit Into Modern EDC Systems
Electronic Data Capture (EDC) systems are comprehensive platforms designed to manage, monitor, and analyze clinical trial data efficiently. Rather than functioning as standalone tools, eCRFs are implemented and governed within the EDC environment. Their effectiveness depends on the broader system’s validation engine, integration framework, audit capabilities, and access controls.
The Role of eCRFs Within an EDC System
Structured Data Capture at the Site Level
eCRFs provide investigators and site staff with a standardized interface to enter participant data according to protocol requirements. Visit structures, form layouts, and field definitions ensure consistency across sites and geographies.
Validation and Data Quality Enforcement
Edit checks, conditional logic, and automated queries are implemented through eCRFs but governed by the EDC’s validation framework. This allows data quality issues to be identified at the point of entry while maintaining centralized oversight and auditability.
System-Level Integration and Data Flow
eCRFs receive data originating from multiple trial systems, including RTSM, ePRO, laboratory platforms, and imaging systems. The EDC manages these integrations, reconciliation rules, and traceability, ensuring that data remains consistent and compliant across sources.
Enablement of Review, Monitoring, and Analysis
Once data is captured through eCRFs, it becomes immediately available within the EDC for centralized review, risk-based monitoring, and medical oversight. Structured eCRF data supports efficient interim analyses, safety evaluations, and progression toward data lock and regulatory submission.
Clarifying Common Misconceptions
EDC Is Not the Same as an eCRF
An eCRF is a core component of an EDC system, but the EDC encompasses broader capabilities such as study configuration, validation management, user roles, audit trails, reporting, and compliance controls.
eCRFs Do Not Operate Independently
Without the EDC framework, eCRFs lack the infrastructure required for secure data storage, controlled integrations, versioning, and regulatory readiness.
Automation Does Not Replace Oversight
While EDC-enabled eCRFs support automated workflows and data checks, clinical judgment from data managers, monitors, and reviewers remains essential to ensure data accuracy and trial integrity.
Understanding how eCRFs function within modern EDC systems is critical for optimizing site workflows, maintaining data quality, and ensuring regulatory compliance in today’s complex clinical trials.
Challenges in eCRF Clinical Trials
Despite widespread adoption, eCRF clinical trials continue to face significant challenges.
- Overly complex and rigid form designs increase site workload and slow data entry, often leading to incomplete, inconsistent, or low-quality data.
- Misinterpretation of protocol requirements during CRF build results in incorrect field definitions, workflows, or visit structures, creating downstream corrections during data review.
- High query volumes caused by weak or misaligned validations that fail to prevent errors at the point of entry, shifting the burden to manual review and query resolution.
- Manual rework during protocol amendments requires repeated updates to eCRFs, increasing the risk of version inconsistencies and operational delays.
- Limited interoperability with RTSM, ePRO, labs, and imaging systems that fragments data flows and complicates reconciliation, traceability, and oversight
Challenge | Consequence |
Inconsistent data entry | Increased query volumes and rework |
Late error detection | Delayed database lock |
Manual reviews | Higher operational burden on data teams |
How AI Is Enhancing eCRF Design and Standardization
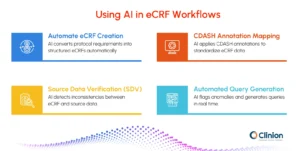
The incorporation of Generative AI (Gen AI) and Artificial Intelligence/Machine Learning (AIML) in eCRFs plays a pivotal and game-changing role in further revolutionizing the effectiveness of eCRFs. Here’s a look at some exciting advancements on the horizon:
Automate eCRF Creation
GenAI streamlines eCRF creation by intelligently analyzing clinical trial protocols, leveraging pre-built global libraries of standardized data fields, and automatically drafting eCRFs. This reduces manual effort, minimizes errors, and saves time.
Mapping of CDASH Annotations
CDASH is a part of the CDISC initiative and guides eCRF development. By mapping CDASH annotations in eCRFs, researchers can ensure that the data is captured in the eCRFs with standardized CDASH terminologies. This, in turn, standardizes the collection, documentation, and reporting of the data and interoperability, thereby facilitating data sharing and regulatory compliance across the research community.
Source Data Verification (SDV)
AIML can be used in the eCRF system to ensure that the data collected for the trial is valid. Using AIML, the information collected from the participants for the clinical trials can be compared to the actual source. Any anomalies or inconsistencies in the data can then be detected. SDV is instrumental in upholding good clinical practice (GCP), which is extremely important for clinical research.
Automate Query Generation
The integration of AI in eCRFs helps in the detection of anomalies. However, it also automates query generation through alerts and notifications to the concerned personnel, which helps streamline the query resolution process and the optimization of trials.
Conclusion
The role of the electronic case report form is changing. As clinical trials generate more data from more sources, eCRFs are no longer evaluated by how well they capture fields, but by how effectively they support downstream decisions.
Future eCRFs will be defined by standards alignment, adaptability to protocol change, and the ability to reduce manual effort across the data lifecycle. The focus will shift from enforcing data entry to enabling consistency, reuse, and earlier confidence in trial data.
In this environment, eCRFs that remain static or overengineered will struggle to keep pace. Those designed with intelligence, interoperability, and regulatory readiness in mind will quietly determine how efficiently trials move from first patient in to final submission.
How Clinion Is Advancing Standards-Ready eCRFs
Clinion’s eCRF approach reflects where clinical data collection is headed: standards-ready by design, not retrofitted later. Its AI-enabled CDASH coding capability automates what has traditionally been a manual, time-intensive step in eCRF development. Once a form design is finalized, CDASH annotations can be generated across pages in a single action, with fields automatically mapped to the appropriate CDASH labels, domains, and annotations. Teams retain full control to review, accept, or refine mappings, combining automation with necessary oversight. This reduces annotation effort, improves consistency, and accelerates downstream data standardization without altering established workflows.

Abriti Rai writes on the intersection of AI, automation, and clinical research. At Clinion, she develops content that simplifies complex innovations and highlights how technology is shaping the next generation of data-driven clinical trials.
FAQS
Frequently Asked Questions
An eCRF is a digital version of the traditional case report form used to collect and manage patient data during clinical trials. It allows study teams to capture data directly into a secure system, enabling real-time validation, analysis, and monitoring while maintaining data integrity and compliance.
Global libraries provide ready-to-use templates built on established standards like CDASH. They help study teams maintain uniformity in data capture and enable faster study setup without compromising compliance or accuracy.
Generative AI interprets study protocols and creates data fields aligned with trial objectives. It removes repetitive setup work, helping data managers focus on study logic and quality rather than manual form design.
CDASH annotations bring standardization to every data field, making it easier to exchange, compare, and analyze data across studies. They also strengthen traceability, which is vital for regulatory submissions and interoperability.
AI and ML identify mismatches between eCRF entries and source data automatically. This helps detect inconsistencies early and ensures that data remains accurate and aligned with Good Clinical Practice standards.
AI continuously reviews trial data to detect irregularities and raise precise queries. This streamlines communication among study teams, shortens resolution cycles, and helps maintain ongoing data quality.
AI is turning eCRFs into intelligent systems capable of predicting data issues, guiding form creation, and learning from past studies. These advancements will help clinical teams manage data more intuitively and make faster, evidence-based decisions.
Clinion integrates AI, ML, and global libraries into its EDC platform to simplify eCRF creation and review. Its technology enables teams to configure studies efficiently, maintain compliance, and deliver high-quality data at every stage.
Still have questions?
Explore how Clinion AI can accelerate your trial – reach out to our team.
Unlock the Future of Clinical Trials with Clinion.
Cut your trial costs by 35% and accelerate your time-to-market by 30%
Compliance
Fully Compliant with Global Standards